Pharma companies move towards systematic access planning approaches: 2021 Access to Medicine Index
Engagement in systematic access planning has increased from one company in 2018 to 40 per cent of companies in the Index this year
A new shift in pharma company policy could shorten the time people in lower-income countries must typically wait for new medicines and vaccines, reports the 2021 Access to Medicine Index. It highlights that eight leading pharma companies are moving to systematically pair candidates in their R&D pipelines with plans for making them accessible in low- and middle-income countries soon after the products are launched onto markets.
The index also points out that advance planning in this way during clinical development can accelerate the speed at which new products become accessible in poorer countries. Access plans can cover activities such as pricing to ensure affordability, registering products for sale in countries facing high disease burdens, and advance licensing agreements that allow manufacturers to start making generic versions.
“Too many people lack access to innovative health products emerging from R&D pipelines. After years of encouraging access planning, we are now seeing a strategic shift in this direction by pharma companies. This could radically change how fast access to new products is achieved – if company leadership is determined to ensure people living in low- and middle-income countries are not last in line,” Jayasree K Iyer, Executive Director of the Access to Medicine Foundation.
Gap between new R&D policy and current performance
Although the systematic approach to access planning is moving into the mainstream, not all late-stage R&D projects are yet supported by an access plan. Overall, 59 per cent of candidates addressing established R&D priorities have evidence of an access plan, compared to 31 per cent of candidates targeting other diseases or product gaps. The company with the most candidates covered by access plans is GSK (80 per cent of projects assessed), followed by Johnson & Johnson, Pfizer, Takeda, Novartis and Sanofi.
In an example of good practice, GSK systematically develops access plans for all projects once phase II clinical trial results are positive. Meanwhile, Pfizer requires access planning to start for all products two years before the product is launched. Takeda’s access plan for its dengue vaccine project includes a commitment to register the vaccine in dengue-endemic countries, voluntary licences and tiered pricing strategies.
There is a backlog when it comes to access to products already on the market. Companies are addressing access for the poor for less than half of the products analysed by the Index. Countries in lower-income tiers are most consistently overlooked. Just 13 per cent of products that must be administered by a healthcare practitioner – such as injectable treatments for cancer – are offered through access strategies in low-income countries. This rises to 26 per cent of self-administered products, which are mainly pills. The Index finds that small numbers of people per country are gaining access through these initiatives, which largely comprise equitable pricing strategies.
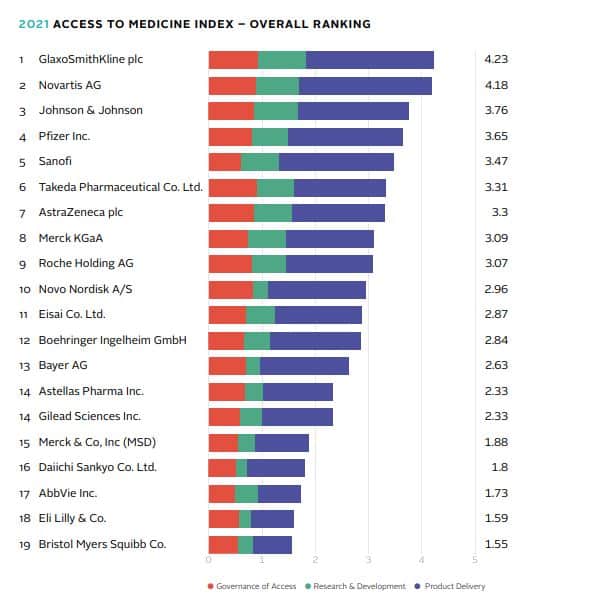
Pfizer joins top 5
The Access to Medicine Index is an independent ranking of 20 of the world’s leading pharma companies on their actions to improve access to medicine in 106 countries. Pfizer is new in the top 5 in 2021, just behind GSK, Novartis and Johnson & Johnson, which hold the top three places. GSK and Novartis are almost tied for first place. The four top-ranked companies together with Sanofi (5th), Takeda (6th), AstraZeneca (7th) and Merck KGaA (8th) comprise the eight companies setting the pace in access planning. Engagement in systematic access planning has increased from one company in 2018 to 40 per cent of companies in the Index.
Although GSK tops the Index, its lead on Novartis is significantly narrower than in 2018. It stands out for its R&D focus and for embedding a clear strategy for addressing access to medicine in its overall corporate strategy. GSK also performs strongly in product delivery, which includes activity in pricing, licensing, donations and capacity building in low- and middle-income countries. However, it has been surpassed in this area by Novartis, which is the only company to demonstrate that it applies equitable access strategies in low-income countries (LICs) for all products analysed, including products from its cardiovascular portfolio. Pfizer has made the most progress in rising from the bottom half of the ranking into the top 5. It owes its rise to a strong performance in access strategies and capacity building as well as access planning in the R&D phase.
R&D in 2020 dominated by cancers and COVID-19
As in 2018, a handful of diseases are the main focus of the pharma companies’ R&D activity. Among infectious diseases, HIV/AIDS, tuberculosis (TB) and malaria once again receive most attention, with COVID-19 newly joining the group in 2020. These diseases account for more than half of projects (198/321 in the communicable disease pipeline. Cancers dominate the pipeline for non-communicable diseases (NCDs), accounting for more than two thirds of such projects (461/663).
R&D activity for COVID-19 patients has been under scrutiny throughout the pandemic. Compared to before the start of the pandemic, the Index finds that the portfolio of experimental drugs and vaccines has filled up for coronavirus patients (from zero to 63 projects), yet remained empty for other pathogens that pose a pandemic risk, such as Ebola, Zika and SARS. Ten of the 16 emerging infectious diseases examined by the Index have empty pipelines from the 20 companies.
The 2021 Index assesses companies on their actions to improve equitable access to products on the market and to make new products available. It covers 82 diseases, conditions and pathogens, including common killers, such as HIV and AIDS, TB and malaria, lower respiratory tract infections, diarrhoeal diseases, maternal health conditions and cancers. People in low- and middle-income countries face more than 80 per cent of the global burden of these diseases. The 20 companies in the Index represent 70 per cent of global pharma revenues.
- Advertisement -



It is a great gesture from all Pharma companies. Thanks for sharing this kinds of articles with us. In this covid-19 time pharma sector is the most essential sector.